Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{Zn}=\dfrac{26}{65}=0,4\left(mol\right)\\ pthh:Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
0,4 0,8 0,4 0,4
\(a,V_{H_2}=0,4.22,4=8,96\left(l\right)\\ b,C\%_{HCl}=\dfrac{0,8.36,5}{150}.100\%=19,5\%\\ c,m_{\text{dd}}=26+150-\left(0,4.2\right)=175,2\left(g\right)\\ C\%_{ZnCl_2}=\dfrac{0,4.136}{175,2}.100\%=31\%\)

a. \(n_{Zn}=\dfrac{6.5}{65}=0,1\left(mol\right)\)
PTHH : Zn + 2HCl -> ZnCl2 + H2
0,1 0,2 0,1
b. \(V_{H_2}=0,1.22,4=2,24\left(l\right)\)
c. \(m_{HCl}=0,2.36,5=7,3\left(g\right)\)
\(n_{Zn}=\dfrac{6,5}{65}=0,1mol\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,1 0,2 0,1
\(V_{H_2}=0,1\cdot22,4=2,24l\)
\(m_{HCl}=0,2\cdot36,5=7,3g\)

a)
$Fe + 2HCl \to FeCl_2 + H_2$
n H2 = n Fe = 11,2/56 = 0,2(mol)
V H2 = 0,2.22,4 = 4,48(lít)
b)
n HCl = 2n Fe = 0,2.2 = 0,4(mol)
=> CM HCl = 0,4/0,4 = 1M
c)
$CuO + H_2 \xrightarrow{t^o} Cu + H_2O$
Ta thấy :
n CuO = 64/80 = 0,8 > n H2 = 0,2 nên CuO dư
Theo PTHH :
n CuO pư = n Cu = n H2 = 0,2(mol)
n Cu dư = 0,8 - 0,2 = 0,6(mol)
Vậy :
%m Cu = 0,2.64/(0,2.64 + 0,6.80) .100% = 21,05%
%m CuO = 100% -21,05% = 78,95%

\(n_{H_2}=\dfrac{6.72}{22.4}=0.3\left(mol\right)\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
\(.........0.6............0.3\)
\(C_{M_{HCl}}=\dfrac{0.6}{0.3}=2\left(M\right)\)
\(n_{Fe_2O_3}=\dfrac{48}{160}=0.3\left(mol\right)\)
\(Fe_2O_3+3H_2\underrightarrow{^{t^0}}2Fe+3H_2O\)
\(1..............3\)
\(0.3..........0.3\)
\(LTL:\dfrac{0.3}{1}>\dfrac{0.3}{3}\Rightarrow Fe_2O_3dư\)
\(n_{Fe}=\dfrac{2}{3}n_{H_2}=\dfrac{2}{3}\cdot0.3=0.2\left(mol\right)\)
\(m_{Fe}=0.2\cdot56=11.2\left(g\right)\)

\(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
\(n_{CuO}=\dfrac{20}{80}=0,25\left(mol\right)\)
PTHH: Zn + 2HCl ---> ZnCl2 + H2
0,1---->0,1----------------->0,1
=> \(\left\{{}\begin{matrix}V_{H_2}=0,1.22,4=2,24\left(l\right)\\C_{M\left(HCl\right)}=\dfrac{0,2}{0,2}=1M\end{matrix}\right.\)
PTHH: CuO + H2 --to--> Cu + H2O
LTL: 0,25 > 0,1 => CuO dư
Theo pthh: nCu = nH2 = 0,1 (mol)
=> mCu = 0,1.64 = 6,4 (g)
\(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\\
pthh:Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,1 0,2 0,2
\(V_{H_2}=0,2.22,4=4,48l\\
C_M=\dfrac{0,2}{0,2}=1M\\
n_{CuO}=\dfrac{20}{80}=0,25\left(G\right)\\
pthh:CuO+H_2\underrightarrow{t^o}Cu+H_2O\\
LTL:0,25>0,1\)
=>CuO dư
\(n_{Cu}=n_{H_2}=0,1\left(mol\right)\\
m_{Cu}=0,1.64=6,4g\)

a, \(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
PTHH: Zn + 2HCl ---> ZnCl2 + H2
0,1--->0,2------->0,1----->0,1
VH2 = 0,1.22,4 = 2,24 (l)
b, \(C_{M\left(HCl\right)}=\dfrac{0,2}{0,2}=1M\)
c, \(C_{M\left(ZnCl_2\right)}=\dfrac{0,1}{0,2}=0,5M\)
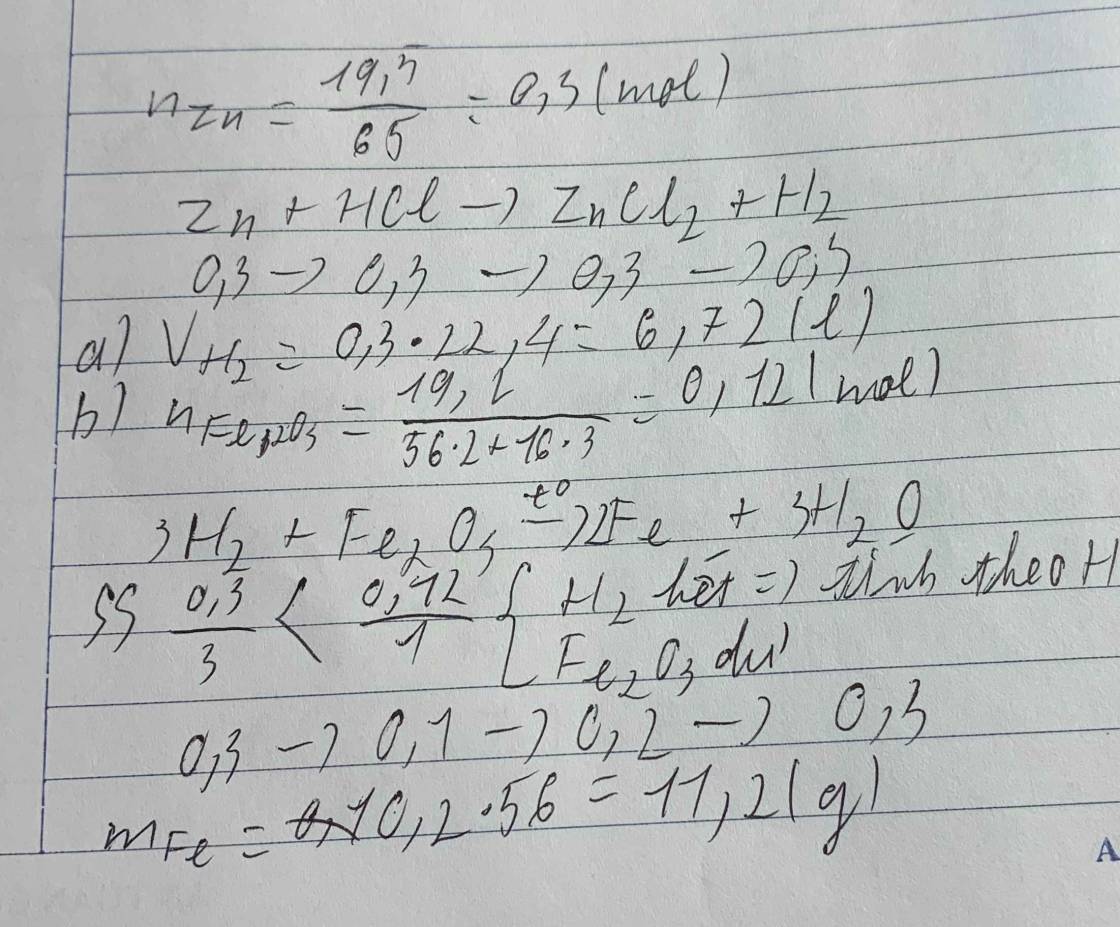

Bài 3: nZn=n/M=6,5/65=0,1(mol)
PT:
Zn + 2HCl -> ZnCl2 + H2\(\uparrow\)
1.......2.............1..............1 (mol)
0,1->0,2 -> 0,1 ->0.1 (mol)
VH2=n.22,4=0,1.22,4=2,24(lít)
b) PT:
H2 + Fe(OH)2 -> Fe + 2H2O
1...........1................1...........2 (mol)
0,1-> 0,1 -> 0,1 -> 0,2 (mol)
=> mFe=n.M=0,1.56=5.6 (g)
Bai 6
Ta co pthh
CuO + H2 \(\underrightarrow{t0}\) Cu + H2O
Theo de bai ta co
nCu=\(\dfrac{48}{80}=0,6mol\)
b,Theo pthh
nCu=nCuO=0,6 mol
\(\Rightarrow\) Khoi luong kim loai thu duoc la
mCu=0,6.64=38,4 g
b,Theo pthh
nH2=nCuO=0,6 mol
\(\Rightarrow\) VH2=0,6.22,4=13,44 l
Bài 5: nFe=m/M=6,5/56=0,116(mol)
VHCl=100ml=0,1 ( lít)
nHCl=CM.V=1.0,1=0,1(mol)
PT:
Fe + 2HCl -> FeCl2 + H2\(\uparrow\)
1..........2..............1.............1 (mol)
0,05 <- 0,1 -> 0,05 -> 0,05 (mol)
VH2=n.22,4=0,05.22,4=1,12(lít)
Chất còn dư là Fe
Số mol Fe dư là 0,116 - 0,1 =0,016 (mol)
Dung dịch sau phản ứng là: \(\left\{{}\begin{matrix}FeCl_2\left(0,05mol\right)\\Fe_{dư}\left(0,016mol\right)\end{matrix}\right.\)
\(C_{M_{FeCl_2}}=\dfrac{n}{V}=\dfrac{0,05}{0,1}=0,5\left(M\right)\)
\(C_{M_{Fe_{dư}}}=\dfrac{n}{V}=\dfrac{0,016}{0,1}=0,16\left(M\right)\)
Bai 1
a,Do tan cua muoi NaCl trong 100g nuoc o nhiet do 200C la
S\(_{NaCl}\) = \(\dfrac{7,18.100}{20}=35,9g\)
b,Theo de bai ta co
mdd=mct + mdm =7,18+20=27,18g
\(\Rightarrow\) C%= \(\dfrac{mct}{mdd}.100\%=\)\(\dfrac{7,18}{27,18}.100\%\approx26,42\%\)
Bài 1:
-Ta có: \(S_{NaCl\left(20^0C\right)}=\dfrac{m_{NaCl}.100\%}{m_{H_2O}}=\dfrac{7,18.100}{20}=35,9\left(g\right)\)
-\(C\%=\dfrac{m_{ct}.100\%}{m_{ct}+m_{dm}}=\dfrac{7,18.100}{27,18}=26,416\left(\%\right)\)
Bài 2: nZn =m/M=6,5/65=0,1 (mol)
VHCl=100ml =0,1 lít
pt:
Zn + 2HCl -> ZnCl2 + H2\(\uparrow\)
1.......2................1..........1 (mol)
0,1->0,2 -> 0,1 -> 0,1 (mol)
Khí sinh ra là H2
VH2=n.22,4=0,1.22,4=2,24(lít)
CM HCl=\(\dfrac{n}{V}=\dfrac{0,2}{0,1}=2\left(M\right)\)
Bai 4:
Ta co pthh
NaOH + HCl \(\rightarrow\) NaCl + H2O
Theo de bai ta co
mNaOH=\(\dfrac{mdd.C\%}{100\%}=\dfrac{200.20\%}{100\%}=40g\)
\(\Rightarrow\) nNaOH=\(\dfrac{40}{40}=1mol\)
Theo pthh
nNaCl=nNaOH=1mol
\(\Rightarrow mNaCl=1.58,5=58,5g\)
mddNaCl= mNaOH + mddHCl = 40 + 100 =140g
\(\Rightarrow\) C% cua NaCl=\(\dfrac{mct}{mdd}.100\%=\dfrac{58,5}{140}.100\%\approx41,79\%\)
b, Theo pthh
nHCl=nNaOH=1mol
\(\Rightarrow\) mHCl=mct=1.36,5=36,5g
\(\Rightarrow\) C%=\(\dfrac{mct}{mdd}.100\%=\dfrac{36,5}{100}.100\%=36,5\%\)
Bài 6: nCuO=m/M=48/80=0,6(mol)
PT:
CuO + H2 -> Cu + H2O
1.............1.........1..........1 (mol)\
0,6 -> 0,6 -> 0,6 -> 0,6 (mol)
mCu=n.M=0,6.64=38,4(g)
VH2=n.22,4=0,6.22,4=13,44 (lít)
Bai 5
Ta co pthh
Fe + 2HCl \(\rightarrow\) FeCl2 + H2
Theo de bai ta co
nFe=\(\dfrac{5,6}{56}=0,1mol\)
nHCl=CM.Vdd=1.0,1=0,1mol
Theo pthh
nFe=\(\dfrac{0,1}{1}mol>nHCl=\dfrac{0,1}{2}mol\)
\(\Rightarrow Fe\) du ( tinh theo so mol cua HCl)
a, Theo pthh
nH2=\(\dfrac{1}{2}nHCl=\dfrac{1}{2}.0,1=0,05mol\)
\(\Rightarrow VH2_{\left(dktc\right)}\)=0,05.22,4=1,12 l
b, Theo pthh
nFe=\(\dfrac{1}{2}nHCl=\dfrac{1}{2}.0,1=0,05mol\)
\(\Rightarrow\) Khoi luong Fe du sau phan ung la
mFe=(0,1-0,05).56=2,8 g
Theo pthh
nFeCl2=\(\dfrac{1}{2}nHCl=\dfrac{1}{2}.0,1=0,05mol\)
\(\Rightarrow CM=\dfrac{0,05}{0,1}=0,5M\)
Bạn thiếu CM của Fe dư
Hậu Trần Công mk bt r cam on ban
nZn=6.5/65=0.1(mol)
a) PTHH: Zn + 2HCl ---> ZnCl2 + H2
(mol) 1 2 1 1
(mol) 0.1 0.1
VH2=0.1x22.4= 2.24 (l)
b) nFe2O3=19.2/160=0.12(mol)
PTHH: Fe2O3 + 3H2 ----> 2Fe + 3H2O
(mol) 1 3 2 3
(mol) 0.1 0.2
Tỉ lệ : 0.12/1 > 0.1/1 =>>> Fe2O3 dư
mFe= 0.2x56=11.2 (g)